Introduction
In Saudi Arabia and across the Gulf region, the pharmaceutical industry is growing rapidly. With this growth comes an unfortunate side effect: a rise in counterfeit medications entering the supply chain. These fake drugs do not just steal money from patients; they can contain harmful ingredients, incorrect dosages, or no active ingredients at all. The result is that people do not get better, conditions worsen, and in severe cases, lives are lost. Handheld Raman analyzers are emerging as powerful weapons in the battle against counterfeit medicines.
In this blog, we will explore how this technology works, why it is so effective, and how pharmaceutical companies and regulators in Saudi Arabia can use it to protect patients.
What is Raman Spectroscopy?
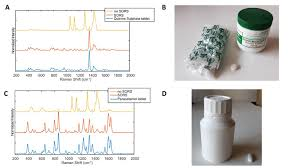
Raman spectroscopy is a way of identifying what something is made of by shining a laser on it and measuring how the light bounces back.
The perfectness of this technology is that it is non-destructive. You do not have to open bottles, dissolve tablets, or send samples to a lab. You can simply point the device at a sealed package and get results in seconds. This makes it perfect for checking medications at warehouses, borders, pharmacies, and hospitals.
Measuring the Impact of Counterfeit Medicines
Counterfeit medicines can include:
- Fake versions of popular brand-name drugs
- Substandard products with incorrect amounts of active ingredients
- Contaminated medications with harmful substances
- Expired drugs repackaged and sold as new
These products look the same as genuine medicines. The packaging, labels, and even the pills themselves can be nearly perfect copies. That is what makes them so dangerous, and why traditional visual inspection isn’t enough anymore.
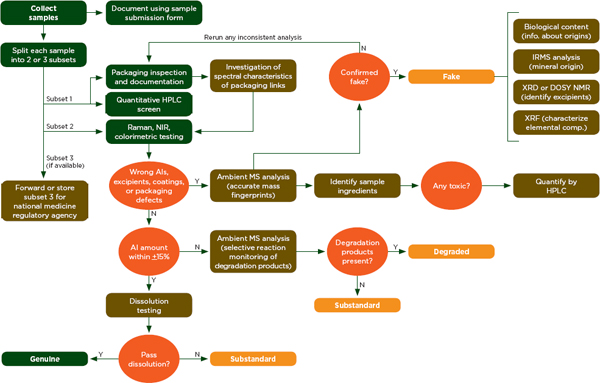
How Raman Technology Detects Counterfeit Drugs
Let’s break down step by step how Raman detects counterfeit drugs and medicines:

Step 1: The Scan
An operator points the handheld Raman analyzer at a medication package or tablet. The device shines a focused laser beam onto the sample. This entire process takes just a few seconds and does not require any sample preparation.
Step 2: Molecular Analysis
The laser interacts with the molecules in the medication. The scattered light creates a unique spectral pattern, that “molecular fingerprint” we talked about earlier. This pattern is immediately captured by the device’s sensor.
Step 3: Database Comparison
The device compares the captured spectrum against a library of known authentic medications. Modern Raman analyzers come pre-loaded with thousands of reference spectra for common pharmaceutical compounds.
Step 4: Instant Results
Within seconds, the device displays a result like “Pass,” “Fail,” or “Unknown.” A “pass” means the medication matches a known authentic product. “Fail” indicates a counterfeit or substandard product. “Unknown” means the substance doesn’t match anything in the database, which is also a red flag.
Benefits of Using Handheld Raman Analyzers in Pharmaceutical Quality Control
Speed and Efficiency
You need to send samples to a lab, wait for technicians to run tests, and then analyze results. With handheld Raman analyzers, you get accurate results in 30 seconds or less. This speed is crucial when you’re screening large shipments or need to make quick decisions at ports of entry.
Non-Destructive Testing
Raman spectroscopy can scan through glass, plastic, and even some blister packs, so you do not have to open the packaging to test the contents. This makes it highly effective for rapid medical testing and pharmaceutical verification without damaging the product. This means:
- Products remain sellable after testing
- No waste or product loss
- Chain of custody is maintained
- Medical testing can be performed anywhere along the supply chain
Cost-Effectiveness
While the initial investment in a handheld Raman device might seem valuable, ranging from $30,000 to $60,000 depending on the model, the long-term savings are important. You can avoid the massive costs associated with counterfeit incidents: recalls, legal issues, and reputation damage.
Portability
These devices are truly portable and can fit in a backpack. This means quality control does not have to happen in a lab. You can take the analyzer to:
- Warehouse receiving docks
- Pharmacy backrooms
- Hospital pharmacies
- Border inspection points
- Distribution centers
- Manufacturing facilities
Minimal Training Required
Most devices are designed with user-friendly interfaces. After a brief training session, which usually requires just a few hours, the staff can confidently perform scans and interpret results.
Comprehensive Detection
Raman spectroscopy provides a complete chemical profile. This means it can identify unexpected contaminants, adulterants, or substitutes that you might not even be looking for.
Best Handheld Raman Devices for Pharmaceutical Quality Control
If you are considering purchasing one for pharmaceutical quality control, here are some of the best handheld Raman devices for pharma QC currently available:
- Thermo Fisher TruScan RM
The TruScan RM is one of the most widely used handheld Raman devices in pharmaceutical quality control worldwide. It’s known for its reliability and extensive pharmaceutical library.
Key Features:
- Scans through clear and amber glass, plastic bottles, and blister packs
- Pre-loaded with an extensive pharmaceutical materials library
- Rapid 30-second analysis
- User-friendly touch-screen interface
- Can create and store custom reference libraries
Best For: Large pharmaceutical companies and regulatory agencies that need a robust, proven solution with strong support.
- Bruker BRAVO
The BRAVO is prized for its fluorescence mitigation technology, which allows it to test substances that would normally interfere with Raman analysis.
Key Features:
- Duo LASER technology reduces fluorescence interference
- Can test colored tablets and formulations
- Compact and lightweight design
- Wireless connectivity for data transfer
- Extensive spectral libraries available
- Works through multiple layers of packaging
Best For: Organizations that need to test a wide variety of formulations, including those with high fluorescence.
- Metrohm MIRA P
The MIRA P is known for its exceptional through-barrier scanning capabilities and ergonomic design.
Key Features:
- Advanced orbital raster scanning (ORS) technology
- Scans through thick and colored containers
- Antimicrobial housing for cleanroom use
- Built-in barcode scanner for sample tracking
- Cloud connectivity for library management
- Highly durable construction
Best For: Pharmaceutical warehouses and distribution centers that need to scan large volumes of products quickly.
- Ocean Insight IDRaman
This device offers excellent value for organizations new to Raman technology or with smaller budgets.
Key Features:
- Affordable entry point into Raman technology
- High-resolution spectral performance
- Lightweight and portable
- Easy-to-use software
- Expandable spectral libraries
- Good battery life for field use
Best For: Small to medium-sized pharmaceutical companies, pharmacies, or hospitals starting their counterfeit detection programs.
Implementing Raman Technology in Your Pharmaceutical Quality Control Process
When you buy handheld Raman analyzer for pharma applications, the purchase is just the first step. Here’s how to successfully implement handheld Raman technology in your quality control workflow:
- Develop Standard Operating Procedures (SOPs)
Create clear, written procedures for when and how to use the Raman analyzer. This should include:
- Which products require testing
- Testing frequency
- How to interpret results
- What to do when a counterfeit is detected
- Documentation requirements
- Maintenance and calibration schedules
- Train Your Team
Invest in comprehensive training for all staff who will use the device. This should cover:
- Basic principles of Raman spectroscopy
- Proper operation of the specific device
- Safety protocols
- Data interpretation
- Troubleshooting common issues
- Documentation and reporting
- Integrate with Existing Systems
Make sure your Raman analyzer works seamlessly with your current quality management systems. Many devices can connect to:
- Laboratory Information Management Systems (LIMS)
- Enterprise Resource Planning (ERP) systems
- Warehouse Management Systems (WMS)
- Electronic batch records
- Build Your Reference Library
While devices come with pre-loaded libraries, you should also build custom reference spectra for:
- Your specific product formulations
- Products from your approved suppliers
- Common counterfeit patterns in your region
- New products as they’re introduced
- Establish Testing Points
Determine critical control points where testing should occur:
- Raw material receiving
- In-process quality checks
- Finished product release
- Warehouse incoming shipments
- Random market surveillance
- Complaint investigations
- Create Response Protocols
Have clear procedures for what happens when a counterfeit is detected:
- Who gets notified?
- How is the product quarantined?
- What documentation is required?
- When should regulatory authorities be contacted?
- How do you trace the source?
- What follow-up testing is needed?
The Regulatory Environment in Saudi Arabia
Saudi Arabia has been proactive in fighting counterfeit medicines with handheld Raman. The Saudi Food and Drug Authority (SFDA) has implemented strict regulations and quality standards for pharmaceuticals entering the Kingdom.
All pharmaceutical products must meet SFDA registration and quality requirements. Importers and manufacturers must have robust quality control systems in place. The SFDA conducts regular inspections and can require additional testing when concerns arise.
How Raman Technology Supports Compliance
Using handheld Raman analyzers demonstrates your commitment to quality and regulatory compliance. It provides:
- Documented evidence of quality control measures
- Rapid response capability when SFDA requests verification
- Traceability through electronic record-keeping
- Risk mitigation by preventing counterfeit products from entering your supply chain
Many pharmaceutical companies in Saudi Arabia are already using this technology as part of their quality assurance programs. It is becoming an expected standard in the industry.
Conclusion
Handheld Raman technology gives pharmaceutical professionals a powerful and practical tool to fight back. They are already protecting patients across the globe, and they can protect yours too.
For organizations in Saudi Arabia, whether you are a pharmaceutical manufacturer, distributor, pharmacy chain, or hospital, investing in this technology is investing in patient safety. It is about ensuring that when someone fills a prescription, they can trust they are getting genuine, effective medication.
Are You Interested in Handheld Raman Technology?
Contact us to learn how these devices work, compare different models, and get expert advice on implementing Raman spectroscopy in your quality control process.
FAQs
1. Can Raman analyzers detect all types of counterfeit medicines?
Raman spectroscopy is very effective. It can easily spot fake medicines with wrong ingredients or incorrect amounts. If a counterfeit is an exact copy, it works best when combined with other quality checks.
2. How long does a scan take?
Most handheld Raman analyzers complete a scan in 30–60 seconds, including data processing and results display.
3. Do I need to open the packaging to scan medicines?
No, Raman devices can scan through glass bottles, plastic containers, and many types of blister packs without opening them.
4. How often do spectral libraries need updating?
Most manufacturers provide library updates quarterly or semi-annually. You should update immediately when new products are released or when new counterfeit patterns are identified in your region.
5. What is the lifespan of a handheld Raman analyzer?
With proper maintenance and regular calibration, these devices last 7 to 10 years or longer. The laser is usually the component with the longest lifespan, often rated for 20,000+ hours of operation.
6. Are there any safety concerns with the laser?
Handheld Raman analyzers use low-power lasers that are generally safe. Operators should never look directly into the laser aperture and should follow the manufacturer’s safety guidelines.
7. What is the main purpose of handheld Raman analyzers in pharmaceuticals?
They help identify counterfeit and substandard medicines quickly by analyzing the chemical composition of drugs without damaging the sample.
8. Why are counterfeit medicines a serious concern?
They may contain harmful substances, incorrect dosages, or no active ingredients, which can worsen health conditions and pose serious risks to patients.
9. How does Raman spectroscopy identify medicines?
It shines a laser on a substance and measures the scattered light to generate a unique molecular fingerprint that identifies the material.
10. Is Raman testing destructive to the product?
No, it is non-destructive and allows testing without opening the packaging or damaging the medicine.
11. Where can handheld Raman analyzers be used?
They can be used in warehouses, pharmacies, hospitals, manufacturing facilities, and border inspection points.
12. Can Raman analyzers scan through packaging?
Yes, they can scan through materials like glass, plastic, and certain blister packs without needing to open them.
13. How do Raman analyzers determine authenticity?
They compare the scanned spectrum with a database of known authentic pharmaceutical compounds.
14. What kind of results does the device provide?
It typically displays results such as pass, fail, or unknown based on how closely the sample matches reference data.
15. What types of counterfeit medicines can Raman detect?
It can detect fake drugs, substandard formulations, contaminated medicines, and repackaged expired products.
16. Do Raman analyzers require lab environments?
No, they are portable and designed for field use, allowing on-site testing without the need for laboratory support.
17. Who can benefit from using handheld Raman devices?
Pharmaceutical manufacturers, distributors, regulators, pharmacies, and hospitals can all benefit from using them.
18. Are handheld Raman analyzers difficult to operate?
No, most devices have user-friendly interfaces and require only basic training to operate effectively.
19. What makes Raman technology suitable for supply chain screening?
Its speed, portability, and ability to test sealed products make it ideal for screening at multiple supply chain points.
20. Can Raman technology detect unknown substances?
Yes, if a substance does not match the reference library, it will be flagged as unknown, which may indicate a risk.
21. Why is building a custom reference library important?
It ensures accurate verification of specific formulations and suppliers relevant to your organization.
22. How does Raman technology support quality assurance?
It provides fast, reliable chemical identification that strengthens quality control and prevents counterfeit entry into the supply chain.
23. Is Raman spectroscopy useful for regulatory compliance?
Yes, it provides documented quality checks and traceability that support regulatory requirements.
24. How can organizations respond to counterfeit detection?
They should follow predefined protocols such as quarantining products, documenting findings, and notifying relevant authorities.
25. Why should pharmaceutical organizations consider adopting Raman technology?
It enhances quality control, supports regulatory compliance, and strengthens protection against counterfeit medicines across the supply chain.
Contact our experts for personalized equipment guidance, training, and SOP support.
Contact:
Email: sales@apex-instrument.com
Mobile/WhatsApp: +971526191767



