Pharmaceutical products contain multiple active ingredients along with excipients like binders, coatings, and stabilizers. During quality control, labs must separate and measure each component accurately so that no compound interferes with another. Quaternary gradient HPLC allows analysts to work with multiple solvents in a single method, which makes it easy to separate compounds that behave differently within the same sample.



In this blog, we will cover how quaternary gradient HPLC works, why it is used for multi-component drug analysis, and what pharma labs in KSA and across the Middle East should focus on when choosing or upgrading their HPLC systems.
What Is Multi-Component Drug Separation in HPLC
When a sample has multiple compounds, each one needs to travel through the column at a different speed so they can be detected separately. This depends upon how each compound interacts with the stationary phase and how the mobile phase pushes it forward. In isocratic methods, the mobile phase stays constant, which works for simple samples but struggles with complex mixtures. Compounds with different properties may not separate properly. This leads to overlapping peaks or long run times.
Gradient elution improves this by changing the mobile phase during the run, usually from more aqueous to more organic conditions. This helps to separate compounds more effectively and brings out those that would otherwise take too long to elute. Quaternary gradient HPLC builds on this by allowing up to four solvents to be used in one method, which gives more flexibility and better control during method development as compared to simple systems.

How Quaternary Gradient HPLC Works in Pharma Analysis
When pharma analysts get to understand how quaternary gradient HPLC works in pharma analysis, this helps them to handle complex methods with better control and accuracy. The system uses a single high-pressure pump along with a low-pressure mixing valve that combines up to four solvents before they enter the pump. The software manages the exact ratio of each solvent throughout the run, creating a controlled gradient that reaches the column.
This setup allows more flexibility in method design. Analysts can adjust different solvents like acetonitrile, methanol, or buffers at specific time points to improve separation. Because of this, how quaternary gradient HPLC works in pharma analysis becomes especially useful for handling complex samples where multiple compounds need to be separated in one run.
In pharmaceutical labs, this approach is commonly used for separating closely related compounds, analyzing drugs along with their impurities, developing stability-indicating methods, and testing combination drug products. Since the system can precisely control solvent mixing without manual preparation, it improves efficiency while maintaining consistent and reliable results.
Pharmaceutical Impurity Profiling With Quaternary HPLC
Pharmaceutical impurity profiling requires accurate separation and detection of all impurities present in a drug product. Quaternary gradient HPLC makes this process more reliable by improving resolution, sensitivity, and consistency in complex analyses.

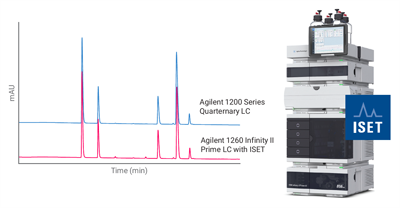

Strict Regulatory Requirements
Pharmaceutical impurity profiling is essential in drug analysis, as regulatory bodies like ICH, FDA, and EMA require the detection and control of impurities above specific limits. Missing or misidentifying an impurity can lead to serious compliance and safety risks.
Better Resolution for Complex Compounds
Quaternary gradient HPLC provides high flexibility in solvent selection, which helps in separating closely related compounds. This is especially useful in pharmaceutical impurity profiling, where degradation products or process impurities may have very similar structures.
Improved Sensitivity at Low Levels
Gradient elution produces sharper and more defined peaks compared to simple methods. This makes it easier to detect and quantify impurities present at very low concentrations, which is critical in regulated pharmaceutical testing.
Faster Analysis with Good Performance
Well-optimized methods on quaternary systems can complete impurity profiling in less time while still maintaining proper separation. This supports higher sample throughput in quality control labs without affecting accuracy.
Consistent and Reproducible Results
Modern quaternary HPLC systems are designed to deliver precise solvent mixing, ensuring consistent results across multiple runs. This reliability is important for maintaining accurate pharmaceutical impurity profiling in regulated lab environments.
The Role of Quality Control Testing in Pharma Labs
Quality control in pharma labs ensures that every drug product meets required standards for safety, quality, and performance. It involves multiple analytical techniques, with chromatography playing a central role in routine testing and release decisions

- Chromatographic methods:They are widely used in quality control testing for drug release and stability studies, especially in HPLC.
- QC teams: They perform key tests such as identity, assay, related substances, dissolution, and content uniformity for finished products.
- Quaternary gradient HPLC: These systems are especially useful for handling complex or multi-component formulations efficiently.
- Method flexibility:QC labs run multiple products on the same system. Quaternary systems allow easy switching between methods using different solvent combinations without changing hardware.
- Regulatory readiness:Methods developed on quaternary systems meet current pharmacopoeial and regulatory expectations. This is important when submitting data to authorities like SFDA and other global agencies.
- Improved efficiency:A single gradient method can analyze assays and related substances together, which reduces the number of runs needed. This saves both time and operational cost in busy QC labs.
How HPLC Supports Pharmaceutical Formulation Analysis
Pharmaceutical formulation analysis involves much more than just identifying the active ingredient. Labs need to accurately measure each drug component in combination products, even when their concentrations vary. It also includes checking excipients that might interfere with drug peaks or degrade over time, which can affect overall product quality.
Also, analysts must monitor trace-level contaminants like leachables from packaging and evaluate drug release behavior in formulations such as coated or modified-release tablets. These tasks require a system that can handle complex samples, maintain consistent gradients, and deliver accurate results across a wide range. Quaternary gradient HPLC is well-suited for this kind of detailed and reliable analysis.
Advantages of Quaternary Systems in Method Development
Quaternary gradient HPLC is especially useful during method development for complex pharmaceutical formulations. It gives analysts more flexibility by allowing multiple solvent combinations instead of relying on just two solvents.
Different solvent ratios, such as acetonitrile and methanol, can be tested within the same system to improve separation. Additional modifiers like formic acid or ammonium formate can be introduced through separate channels for better control.
It also allows blending of different buffer systems to fine-tune selectivity and improve peak resolution. This flexibility reduces the number of experiments needed to find the right mobile phase conditions.
Compared to binary systems, method optimization can be completed much faster with fewer trial-and-error steps. Faster method development helps pharma labs speed up overall workflows and supports quicker product development timelines.
Choosing the Right HPLC System for Your Pharma Lab
Selecting the right HPLC system for a pharma lab involves more than picking a brand. The system needs to meet regulatory requirements, match the lab’s method complexity, and be supported by a supplier who understands the pharma environment. The key considerations include the following:
- Pump performance specifications:Look at gradient accuracy, flow rate range, and pressure capacity. For gradient methods at high flow rates or with fast analysis requirements, pump quality directly affects data quality.
- Detector compatibility:UV/Vis detection is standard for most pharmaceutical methods, but some labs also need PDA capability for peak purity assessment or confirmation of peak identity during impurity profiling.
- System qualification support:Regulatory labs need IQ/OQ/PQ documentation for every instrument. Ensure your supplier can provide this or support a third-party qualification process.
- Software compliance:In regulated pharma environments, the data system must meet 21 CFR Part 11 or equivalent requirements for electronic records and audit trails.
- After-sales service:A quaternary HPLC system is a significant investment. Local technical support, access to spare parts, and preventive maintenance contracts are non-negotiable in a production-critical lab environment.
HPLC Systems for Pharmaceutical Quality Control in Saudi Arabia
The pharmaceutical sector in Saudi Arabia has grown significantly over the past decade, with local manufacturers and contract labs all needing to meet international quality standards. HPLC systems for pharmaceutical quality control in Saudi Arabia are now a central part of lab infrastructure across the country.
Labs regulated by SFDA follow guidelines that align closely with ICH requirements, which means the analytical methods and instruments used must meet international standards. Quaternary gradient systems from established manufacturers meet these standards and are accepted across all major regulatory frameworks.
Whether the lab is handling generic drug development, stability testing, or release testing for regulated markets, the right HPLC system setup makes a measurable difference in data quality and lab efficiency.
Why Work With a Reliable Supplier in the Region
For labs in KSA and across the Middle East, working with a laboratory analytical instruments supplier in the Middle East who truly understands the local regulatory environment and lab challenges matters more than most people realize.
At Apex Instrument, we are a trusted pharmaceutical HPLC instruments supplier in KSA, supporting pharma labs and research institutions with high-performance HPLC systems and related analytical instruments. As an HPLC system supplier in Riyadh, Saudi Arabia, we provide local installation, calibration, operator training, and ongoing technical support to keep your system performing at its best.
We understand what it takes to keep an HPLC running reliably in a regulated pharmaceutical environment, and we stock the consumables and spare parts our clients need for day-to-day operations.
Are You Ready to Upgrade Your HPLC Capabilities?
If your lab is dealing with complex drug formulations, inconsistent separations, or growing regulatory pressure, it’s time to move to a system that actually keeps up.
Apex Instrument helps pharma labs across Saudi Arabia and the Middle East implement high-performance quaternary gradient HPLC systems that deliver precise, reliable, and compliant results.
From system selection to installation, IQ/OQ/PQ qualification, and ongoing technical support, you get everything you need to run your lab without interruptions.
FAQs
1. What is quaternary gradient HPLC, and how is it different from binary HPLC?
Quaternary gradient HPLC uses up to four solvent channels that can be blended in any programmed proportion during a run, whereas binary HPLC uses only two. This gives quaternary systems far more flexibility for complex pharmaceutical formulation analysis and multi-component separations.
2. How does quaternary gradient HPLC help with HPLC drug mixture separation?
Quaternary gradient HPLC improves HPLC drug mixture separation by allowing more precise control over mobile phase composition throughout the run.
3. What is multi-component drug separation in HPLC, and when is it needed?
Multi-component drug separation in HPLC refers to the simultaneous identification and quantification of two or more active pharmaceutical ingredients or related compounds in a single analytical run. It is needed for combination drug products, impurity profiling, and stability-indicating method development.
4. How does quaternary gradient HPLC work in pharma analysis for impurity testing?
By programming a precise solvent gradient using up to four channels, analysts can resolve trace impurities that are structurally similar to the drug substance.
5. What role does pharmaceutical impurity profiling play in drug product approval?
Pharmaceutical impurity profiling is a regulatory requirement under ICH Q3B guidelines. Any impurity above identification or qualification thresholds must be characterized.
6. How does quality control testing in pharma labs use quaternary gradient HPLC?
Quality control testing in pharma labs uses quaternary gradient HPLC for assay testing, related substances analysis, dissolution testing, and uniformity of content. The gradient capability ensures that complex samples are fully resolved within a single analytical run.
7. What is pharmaceutical formulation analysis, and what does HPLC measure in it?
Pharmaceutical formulation analysis is the process of testing a drug product to confirm identity, strength, purity, and performance. HPLC measures the concentration of each active ingredient, identifies impurities, and monitors degradation products in the formulation.
8. Why is gradient elution preferred over isocratic methods for multi-component samples?
Gradient elution is preferred because fixed mobile phase conditions in isocratic methods cannot efficiently resolve compounds with very different polarities in one run.
9. What types of pharmaceutical products benefit most from quaternary gradient HPLC?
Combination tablets, extended-release formulations, injectable products with multiple APIs, and complex biologics intermediates all benefit from quaternary gradient HPLC.
10. How does pharmaceutical impurity profiling differ between development and QC stages?
During development, pharmaceutical impurity profiling involves identifying all unknown degradation products and process impurities. In QC, the method is already validated, and the focus shifts to routine quantification of known impurities against established limits.
11. What should I look for in HPLC systems for pharmaceutical quality control in Saudi Arabia?
When evaluating HPLC systems for pharmaceutical quality control in Saudi Arabia, look for systems that meet ICH and SFDA analytical requirements, come with IQ/OQ/PQ qualification support, use data systems compliant with electronic records regulations, and are backed by a local service team.
12. How does a quaternary pump improve method development efficiency?
A quaternary pump allows method developers to explore combinations of up to four solvents, which compresses the experimental timeline.
13. Is quaternary gradient HPLC suitable for stability-indicating methods?
Yes, quaternary gradient HPLC is widely used for stability-indicating method development because it provides the resolution power needed to separate a drug from all its degradation products.
14. What detectors are commonly used with quaternary gradient HPLC in pharma?
UV/Vis and photodiode array (PDA) detectors are the most commonly used with quaternary gradient HPLC in pharma.
15. How does a laboratory analytical instrument supplier in the Middle East support pharma labs?
A laboratory analytical instruments supplier in the Middle East provides calibration services, operator training, validation documentation, and access to certified consumables and spare parts.
16. What are the regulatory requirements for HPLC methods used in QC testing in Saudi Arabia?
QC methods used under SFDA oversight must be validated per ICH Q2(R1) guidelines, covering specificity, linearity, precision, accuracy, range, limit of detection, and limit of quantitation.
17. Can quaternary gradient HPLC handle both small molecules and large biologics?
Yes, quaternary gradient HPLC handles small molecule drugs very well and is also used for peptide analysis, oligonucleotides, and some protein characterization applications, depending on the column and detection method.
18. What is the benefit of using a quaternary HPLC for dissolution testing in pharma labs?
Dissolution testing combined with HPLC analysis is an important part of quality control testing in pharma labs for oral dosage forms. A quaternary gradient system can run dissolution samples from combination products and resolve all active ingredients in a single analytical sequence, which improves throughput compared to running separate methods.
19. How do I know if my HPLC system needs to be upgraded to a quaternary gradient setup?
If your lab is developing methods for combination products, struggling to resolve complex matrices with a binary or isocratic system, or needs faster analytical throughput without compromising resolution, then your HPLC system may need to be upgraded to a quaternary gradient setup.
20. Why is it important to work with a pharmaceutical HPLC instrument supplier in KSA?
Working with a pharmaceutical HPLC instruments supplier in KSA means you get access to local technical expertise, faster response times for service calls, and a supplier who understands the regulatory environment in which your lab operates.
21. What kind of training is needed to operate a quaternary gradient HPLC system in a pharma lab?
Analysts need training on gradient method programming, solvent preparation, system startup and shutdown, routine maintenance tasks, and troubleshooting.
22. How does column selection affect quaternary gradient HPLC performance for drug separation?
Column chemistry, particle size, and pore size all influence how well compounds separate in HPLC drug mixture separation.
23. What is the difference between assay testing and related substances testing in pharma HPLC?
Assay testing quantifies the amount of active drug in a formulation, while related substances testing identifies and quantifies impurities and degradation products.
24. How does Apex Instrument support HPLC system qualification in Saudi Arabia?
As a pharmaceutical HPLC instruments supplier in KSA and an HPLC system supplier in Riyadh, Saudi Arabia, Apex Instrument supports instrument qualification through IQ/OQ/PQ documentation, calibration services, and ongoing performance verification to help labs meet both SFDA and international regulatory requirements.
25. What are the most common mobile phase combinations used in quaternary gradient HPLC for pharma?
Common mobile phase combinations include acetonitrile-water with an aqueous buffer, methanol-acetonitrile blends with ammonium formate, or ternary combinations using a pH modifier as a third channel.
26. Why is data integrity important in pharmaceutical HPLC systems?
HPLC chromatography data systems must maintain complete, accurate, and unalterable records of all analytical results. Without this, data submitted for product approval or batch release is not acceptable to regulatory authorities.
27. How does a laboratory analytical instruments supplier in the Middle East handle after-sales service?
A capable laboratory analytical instruments supplier in the Middle East provides scheduled preventive maintenance, rapid response to breakdown calls, genuine spare parts, and remote diagnostics.
28. What is the typical validation process for a quaternary gradient HPLC method in pharma?
Method validation involves demonstrating specificity, linearity, accuracy, precision, robustness, and limits of detection and quantitation per ICH Q2(R1).
29. Can quaternary gradient HPLC be used for raw material testing in pharma?
Yes. Quaternary gradient HPLC is used for raw material identification and purity testing in pharma, including testing of starting materials, intermediates, and excipients.
30. How can pharma labs in KSA get access to advanced quaternary HPLC systems and technical support?
Pharma labs in KSA looking for advanced quaternary HPLC systems can reach out to Apex Instrument, an established pharmaceutical HPLC instruments supplier in KSA and trusted laboratory analytical instruments supplier in the Middle East.

Contact our experts for personalized equipment guidance, training, and SOP support.
Contact:
Email: sales@apex-instrument.com
Mobile/WhatsApp: +971526191767



