Quaternary gradient HPLC troubleshooting is important for maintaining consistent performance in pharmaceutical laboratories. These systems are precise, powerful, and incredibly sensitive. Problems like worn pump seals, contaminated solvents, or loose fittings can quickly disrupt a run. They are commonly used for method development, impurity profiling, assay testing, and stability studies. This added flexibility increases system complexity, which makes regular maintenance, proper setup, and a structured troubleshooting approach necessary for reproducible analytical results.
This blog will cover the most common challenges in HPLC systems and give you beneficial and step-by-step solutions so that you can apply them later.

How Quaternary HPLC Systems Work
Before looking at quaternary HPLC problems and their solutions, it is necessary to first understand how these systems are different from binary or isocratic setups.
A quaternary pump uses a single high-pressure pump with a low-pressure mixing valve that blends up to four solvents before the pump. This design is efficient and cost-effective, but the mixing happens before pressurization. That means any issue with solvent quality, degassing, or valve performance will directly affect the pump output and the separation. Pharma labs depend upon this setup for gradient methods because it gives more control over mobile phase composition. But that control is only reliable when the system is well-maintained and properly set up.
Why Baseline Drift Happens in HPLC Systems
Baseline drift in HPLC systems is a common issue in pharma labs and usually appears as a slow rise or fall in the baseline during a run in gradient methods. Below are the root causes that help in fixing the problem quickly and maintaining results.
Incomplete Mobile Phase Equilibration
If the system and column are not fully stabilized at the starting conditions, baseline drift in HPLC systems can occur. Starting a run too early often leads to unstable results. It is important to give the system enough time to equilibrate properly before beginning any analysis.

Solvent Quality Issues
The quality of solvents directly affects baseline stability. Low-grade acetonitrile or methanol may absorb UV light at certain wavelengths, causing fluctuations. Using high-purity, consistent-grade solvents helps maintain a stable baseline and reliable results.
Temperature Variations
Small changes in lab temperature can impact detector response and overall baseline stability. Sensitive detectors, especially refractive index-based ones, are more affected by temperature shifts, making controlled lab conditions important.
Column Bleed
Older or worn-out columns can release small amounts of the stationary phase during gradient runs. This can lead to gradual baseline changes. Running a blank gradient is a simple way to check if the column is causing the issue.
Aging UV Lamp
A UV lamp that is nearing the end of its life may produce inconsistent light output. This can result in baseline instability. Regular monitoring of lamp usage hours and timely replacement helps ensure consistent detector performance.
What Causes Pressure Drop in HPLC
When your system pressure drops unexpectedly, it usually points to one of a few things. What causes pressure drop in HPLC is a question that comes up often, and the answer is almost always tied to one of these areas:
- A leak in the flow path: Even a small leak at a fitting or connection will cause pressure loss. Go through each connection methodically and check for moisture or solvent residue.
- A worn pump seal or piston: Pump seals wear over time, especially in labs running aggressive solvents. A damaged seal will not hold pressure properly, and you will notice the pressure fluctuate rather than hold steady.
- Air bubbles in the system: Gas trapped in the pump or tubing will compress instead of pushing solvent, causing pressure instability. Make sure all solvents are properly degassed and that the priming procedure was done correctly.
- A partially blocked check valve: Check valves control the direction of solvent flow in the pump. If they are contaminated or worn, they allow backflow, which disrupts the pumping cycle and causes pressure irregularities.
- Blockage in the inline filter or guard column: If the pressure is consistently low rather than fluctuating, look at the filters upstream of the column. Replace the inline frit or guard column as needed.

Common HPLC System Problems in Pharma Labs
Beyond baseline drift and pressure issues, pharma labs deal with a wider range of common HPLC system problems in pharma labs that affect data quality and instrument uptime.
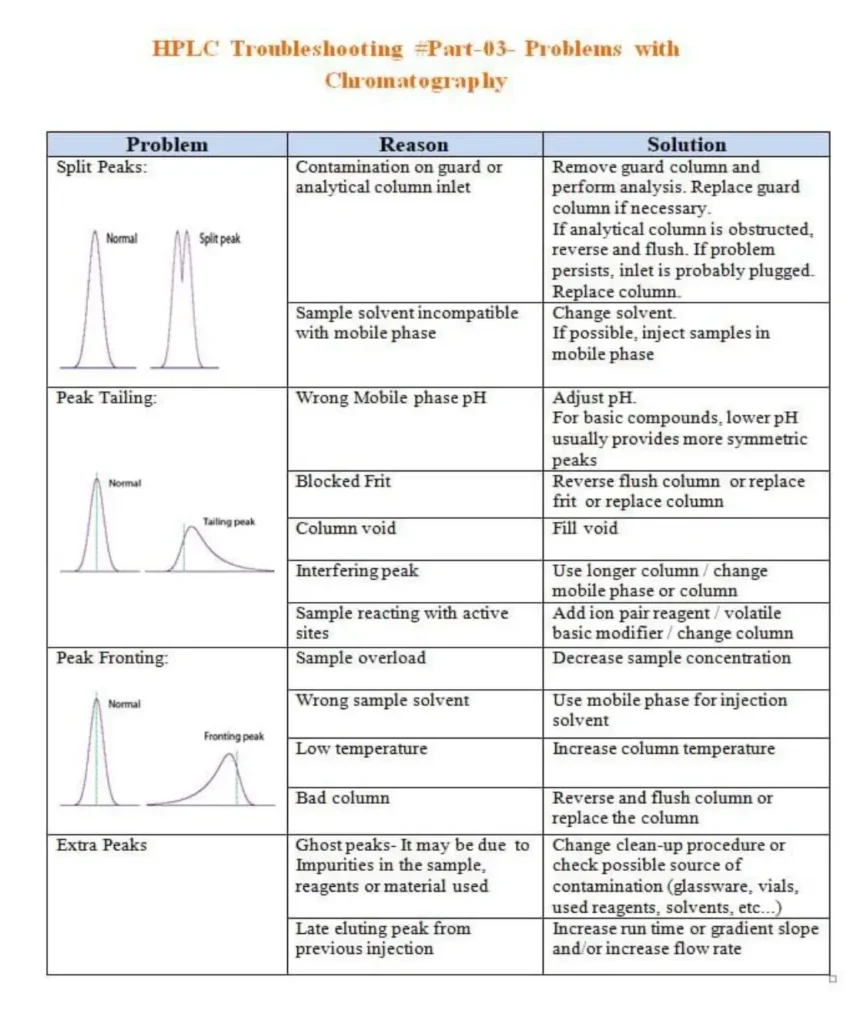
Peak Splitting or Shouldering
This usually points to a void at the head of the column. It can also result from a sample that is not fully dissolved in the mobile phase. Always dissolve samples in the weakest solvent in your gradient.
Ghost Peaks or Extra Peaks
These often come from contaminated autosampler wash solvents, leftover samples from a previous injection, or a contaminated mobile phase. Clean the autosampler thoroughly and refresh your solvents.
Poor Retention Time Reproducibility
If your retention times shift from run to run, check column temperature stability, flow rate accuracy, and mobile phase preparation consistency. Even minor changes in solvent ratio affect retention.
Tailing Peaks
Peak tailing in reversed-phase methods often means the column is overloaded, or there is a secondary interaction with residual silanol groups on the stationary phase. Reduce sample concentration or switch to a column with end-capped silica.
Weak Signal
Check detector settings, wavelength selection, and lamp condition. Also, confirm the sample was prepared at the correct concentration and that the injection volume is set properly.

How to Troubleshoot HPLC System Step by Step
To know how to troubleshoot HPLC step by step, the following steps are a structured approach that works across most scenarios:
Step 1 – Define the problem clearly: Note what is different today versus the last successful run. There are some questions, like Pressure? Baseline? Peak shape? Retention time? Being specific saves time.
Step 2 – Check recent changes: It involves some queries. Was a new column installed? Did you change a lot of solvent? Was any maintenance done? Changes often cause problems that do not show up immediately.
Step 3 – Run a system suitability test: System suitability testing in pharma labs is designed exactly for this test. Use a reference standard and check plate count, tailing factor, and resolution against your method’s acceptance criteria. This tells you if the issue is with the instrument or the chemistry.
Step 4 – Isolate the problem: Replace one component at a time. Swap columns, change solvents, or bypass the autosampler to narrow down the source.
Step 5 – Check the pump: Verify flow rate accuracy using a volumetric check. Watch pressure stability over time. A fluctuating baseline pressure often points to pump issues.
Step 6 – Inspect fittings and connections: Finger-tighten, then use a wrench just enough to stop any rotation. Over-tightened fittings strip easily and can damage ferrules permanently.
Step 7 – Document everything: Good troubleshooting logs make future problems easier to solve and provide a clear audit trail for regulatory purposes.
System Suitability Testing in Pharma Labs
System suitability testing in pharma labs is a useful checkpoint that tells you if your HPLC system is ready to produce reliable data.
Parameters like theoretical plates, tailing factor, resolution, and relative standard deviation of peak area give you a snapshot of system performance at the start of each analytical sequence in pharmaceutical laboratories. If any parameter falls outside the acceptance criteria in your method, you stop and investigate before wasting time on actual samples.
For quaternary gradient systems, system suitability runs should also verify gradient accuracy. Some labs use dye tests or UV-absorbing markers to confirm that the pump is delivering the correct solvent proportions at each gradient step. Regular system suitability testing also extends instrument life by catching early signs of wear before they become serious failures.
Quaternary Gradient HPLC Troubleshooting for Gradient-Specific Issues
Some problems are unique to gradient methods and do not show up in isocratic runs. Here is what to watch for in quaternary gradient HPLC troubleshooting:
- Gradient dip or artifact peaks at early time points: These often come from solvent impurities or UV-absorbing additives in one of the mobile phase components. Use the highest purity solvents available and run a blank gradient as part of every analytical sequence.
- Inconsistent gradient shape: If the proportional valve in the quaternary pump is not working correctly, the actual gradient delivered will not match what you programmed. This leads to inconsistent retention and peak areas. Verify gradient accuracy with a photometric test.
- Carryover between solvents: When switching between very different solvent compositions, flushing the system thoroughly matters. Incomplete flushing leads to mobile phase mixing errors that affect the first few runs of a sequence.
- Slow equilibration: Quaternary systems sometimes take longer to equilibrate between runs because of the low-pressure mixing design. Build enough equilibration time into your sequence, especially for methods that return to high aqueous conditions.
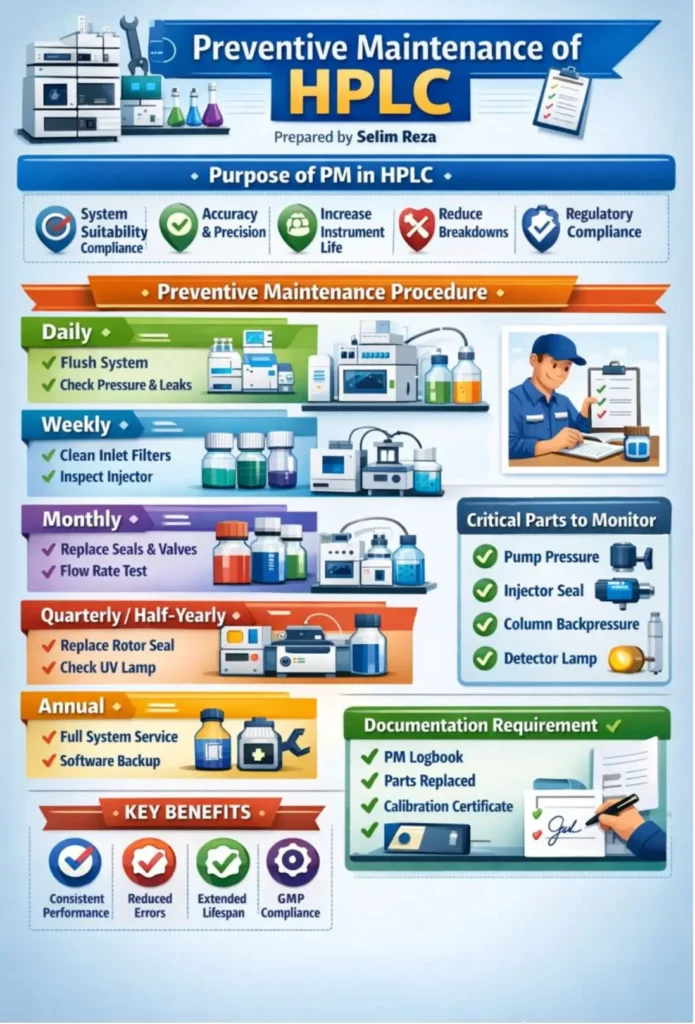
How Regular Maintenance Prevents HPLC Issues
Most quaternary HPLC problems and solutions are closely linked to how well the system is maintained. Labs that follow a proper maintenance routine usually face fewer unexpected issues and less downtime. It is always better to replace pump seals on time instead of waiting for failure. Flushing the system at the end of each day helps remove any buildup and keeps the flow path clean.
Regularly checking the autosampler needle and rotor seal also prevents performance issues. Inline frits should be replaced when you notice a gradual rise in pressure. Keeping a record of column usage and condition helps track performance over time. These simple practices improve reliability, protect data quality, and extend the life of the system.
Why Supplier Support is Important in KSA Pharma Labs
Pharma labs in Saudi Arabia work under strict regulatory requirements. When an HPLC system stops working, it can delay analysis and impact timelines. This is why partnering with an experienced advanced quaternary HPLC systems supplier in KSA is important. They can provide timely support and help restore system performance quickly.
At Apex Instrument, we supply and support high-performance analytical instruments across KSA, including quaternary HPLC systems from trusted manufacturers. If you need on-site support, spare parts, or system calibration, then our HPLC troubleshooting services in Saudi Arabia are designed to minimize downtime.
As a pharma laboratory analytical instruments supplier, we understand what pharma labs need beyond just the instrument itself. Reliable after-sales service, technical training, and access to certified consumables are all part of what we bring to our clients. If you are looking for an HPLC system supplier in KSA or need support with an existing system, we are here to assist you.

Get Your Quaternary HPLC System Back to Peak Performance
If recurring HPLC issues are slowing your lab down, it’s time to stop troubleshooting blindly and start fixing problems with expert support from Apex Instruments that understands pharma workflows.
From baseline drift to pressure instability, every minute of downtime impacts your results, timelines, and compliance. Do not let system issues compromise your analysis. Get the support your lab actually needs.
Request A Quote
FAQs
1. What is quaternary gradient HPLC troubleshooting, and why does it matter in pharma labs?
Quaternary gradient HPLC troubleshooting is the process of identifying and fixing performance issues in HPLC systems that use up to four solvents in a gradient program. It matters because even small issues can compromise analytical results, which directly affect product quality decisions in pharmaceutical manufacturing.
2. What are the most common quaternary HPLC problems and solutions in day-to-day lab work?
The most common quaternary HPLC problems and solutions involve baseline drift, pressure instability, poor peak shape, and retention time variability.
3. What causes baseline drift in an HPLC system during a gradient run?
Baseline drift in an HPLC system is most caused by incomplete equilibration, solvent impurities, UV-absorbing contaminants in the mobile phase, or an aging detector lamp.
4. How does system suitability testing in pharma labs help catch HPLC problems early?
System suitability testing in pharma labs checks key performance parameters like plate count, tailing factor, and resolution before any sample is analyzed.
5. How do I troubleshoot an HPLC system step by step without wasting time?
To troubleshoot an HPLC system step by step, start by clearly defining the symptom. Then check for recent changes, run a system suitability test, isolate the problem to a specific component, and replace or adjust one thing at a time while documenting each step.
6. What causes pressure drop in HPLC and how should I respond?
The cause of pressure drop in HPLC is usually a leak, a worn pump seal, an air bubble, a faulty check valve, or a blocked filter. You should start with a visual inspection of all fittings, then move to the pump components if there is no external leak is found.
7. Are common HPLC system problems in pharma labs different from those in other industries?
The core problems are similar, but pharma labs face stricter documentation and regulatory requirements. Common HPLC system problems in pharma labs need to be resolved with validated methods and a clear audit trail.
8. How often should system suitability testing in pharma labs be performed?
System suitability testing in pharma labs should be performed at the beginning of every analytical sequence involving regulated assays.
9. What are the signs of a failing pump seal in a quaternary HPLC system?
Signs include a gradual pressure drop, baseline noise that corresponds to the pump cycle, and solvent leakage around the pump head.
10. Can baseline drift in HPLC be caused by the column alone?
Yes, baseline drift in an HPLC system can come from column bleed, especially with older or degraded columns during high-organic gradient steps.
11. How do I verify gradient accuracy in a quaternary HPLC system?
Gradient accuracy is verified using a photometric test with a UV-absorbing marker at a known wavelength. This confirms that the pump is delivering the correct solvent proportion at each step.
12. What role does solvent quality play in quaternary HPLC problems and solutions?
Solvent quality plays an important role. Low-grade solvents introduce UV-absorbing impurities and dissolved gases that create noise, drift, and pressure instability. Always use HPLC-grade or LC-MS-grade solvents, especially for UV-sensitive methods.
13. What is the best way to eliminate air bubbles in a quaternary pump?
Proper degassing is the best way. Use an online degasser if your system has one, or degas manually by sonication under vacuum. If bubbles persist, check for micro-leaks at fittings, since air can enter even when solvent is not leaking out.
14. How does temperature affect baseline drift in HPLC systems?
Temperature changes affect solvent viscosity, refractive index, and column selectivity. Baseline drift in an HPLC system is often temperature-related in labs without climate control.
15. What should I check first when retention times start shifting between runs?
Check mobile phase preparation and flow rate accuracy first. Retention time shifts are one of the most common HPLC system problems in pharma labs and are often traced back to small variations in solvent ratio or column temperature from one run to the next.
16. Is there a standard protocol for how to troubleshoot an HPLC system step by step?
Most regulatory guidance documents and instrument manufacturers recommend a structured approach: document the symptom, review recent changes, run a suitability test, isolate components, and fix one issue at a time.
17. What causes a pressure drop in HPLC when the system has recently been serviced?
After servicing, what causes pressure drop in HPLC is usually an improperly seated fitting, a new seal that needs seating time, or residual air from the service procedure.
18. How do I distinguish between a detector problem and a mobile phase problem?
Inject a fresh reference standard with a known mobile phase from a trusted lot. If the detector response is still poor, the issue is likely with the detector or its lamp. If results improve, the original mobile phase was the problem.
19. Can poor sample preparation cause what looks like an HPLC system failure?
Yes, it can. Ghost peaks, poor peak shape, and carryover are among the common HPLC system problems in pharma labs that actually originate from sample preparation.
20. How do I know when a column needs to be replaced rather than reconditioned?
If back pressure is high even after flushing, if resolution no longer meets system suitability criteria, or if peak shape has permanently deteriorated despite cleaning, the column has reached the end of its useful life and should be replaced.
21. What is the impact of baseline drift in HPLC on quantitative accuracy?
Baseline drift in an HPLC system affects integration accuracy, especially for late-eluting peaks or impurities near the baseline. Even small drifts can lead to over- or under-reporting of peak areas.
22. How does system suitability testing in pharma labs connect to regulatory compliance?
System suitability testing in pharma labs is required by pharmacopoeias, which include the USP and EP. Regulatory agencies expect to see suitability data as part of the analytical sequence, and any deviation must be documented and investigated before results are reported.
23. What are the signs of a failing check valve in a quaternary pump?
Fluctuating baseline pressure, irregular flow delivery, and backflow are all signs of a failing check valve in a quaternary pump.
24. What does an advanced quaternary HPLC systems supplier in KSA actually support?
A good advanced quaternary HPLC systems supplier in KSA should offer installation, calibration, operator training, preventive maintenance contracts, and access to certified spare parts and consumables.
25. Can HPLC troubleshooting services in Saudi Arabia help with method validation issues?
Yes, HPLC troubleshooting services in Saudi Arabia from an experienced supplier can help identify if method performance issues come from instrument condition, column selection, or method design.
26. What makes a pharma laboratory analytical instruments supplier different from a regular equipment dealer?
A pharma laboratory analytical instruments supplier understands regulatory requirements, validation documentation, and the specific performance standards that pharma labs must meet. They offer technical guidance, calibration support, and compliance-ready documentation.
27. How do I choose an HPLC system supplier in KSA for a regulated pharma lab?
You can look for an HPLC system supplier in KSA that has a track record with pharma clients, can provide IQ/OQ/PQ qualification support, offers local after-sales service, and carries instruments from manufacturers with established regulatory acceptance.
28. What is the first thing to check when a quaternary HPLC run produces no peak at all?
You can check the detector wavelength setting, the sample concentration, and if the injection actually took place. Then, verify that the mobile phase is flowing by watching the pressure reading.
29. How do reagent or buffer preparation errors show up in HPLC results?
They appear as shifts in retention time, poor peak shape, or unexpected baseline behavior. Errors in buffer pH or concentration are a frequently overlooked source of quaternary HPLC problems and solutions that analysts sometimes attribute to the instrument when the issue is actually in the chemistry.
30. Where can pharma labs in Saudi Arabia find reliable HPLC support and instruments?
For reliable HPLC troubleshooting services in Saudi Arabia and access to advanced quaternary HPLC systems, pharma labs can reach out to Apex Instrument.



